Mr. H then told us to get out our review packet and turn to page 19 which was the density reading sheet. Since we had already gone over problems 1-6, we only went over problems 7-14. The answers for these problems are:
7. C; temperature
8. A; increases
9. B; 63 degrees
10. B; 35 grams
11. B; they absorb certain colors of light and reflect the rest to our eyes.
12. D; bluish-green
13. D; 820nm
14. C; reddish-orange
Mr. H told us while we were writing down the answers that these types of questions would be on our ACT. Some questions would be easier like question 7, while questions like 14 would be more challenging.
After we wrote our answers down, Mr. H reviewed temperature and accuracy vs. precision. The three major temperature scales are: Fahrenheit, Celsius and Kelvin. The formulas for finding temperatures are: *F=1.8*C+32, *C=(*F-32)/1.8 and K=273.15+*C. The boiling point of these temperatures are: 212*F, 100*C and 373.15K. The freezing point are: 32*F, 0*C and 273.15K. For additional reference, use page 8 of the book. Mr. H showed us a picture of three dartboards with one portraying inaccu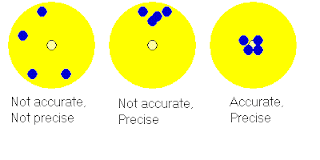 rate and imprecise throws, the other portraying accurate, but imprecise throws, and the last one portraying both accurate and precise throws. Here is a picture relating to this idea.
rate and imprecise throws, the other portraying accurate, but imprecise throws, and the last one portraying both accurate and precise throws. Here is a picture relating to this idea.
 rate and imprecise throws, the other portraying accurate, but imprecise throws, and the last one portraying both accurate and precise throws. Here is a picture relating to this idea.
rate and imprecise throws, the other portraying accurate, but imprecise throws, and the last one portraying both accurate and precise throws. Here is a picture relating to this idea. After this, Mr. H reviewed the classifications of matter on page 16. The answers are as follows:
A. HOM
B. HEM
C. E/C
D. HEM
E. HOM
A. C
B. E
C. C
D. HOM
E. HOM
F. HOM
G. HEM
H. HEM
I. HEM
J. HOM
A. F
B. F
C. F
D. T
E. F
F. T
G. F
Some key classification of matter terms are element, compound, homogeneous mixtures and heterogeneous mixtures.
Element- Type of matter that cannot be broken down into two or more pure substances. Ex. Oxygen
Compound- A pure substance that contains more than one element. Ex. Water is a compound of oxygen and hydrogen.
Homogeneous mixture- The composition is the same throughout, also known as a solution. Ex. brass is a homogeneous mixture of zinc and copper.

Heterogeneous mixture- The composition varies throughout. Ex. Granite contains discrete regions of different mixtures (feldspar, mica, and quartz).

After we finished reviewing, Mr. H told us to finish our Beverage Lab. Two group members of each group went to the computer lab to graph the data while the other group members found and recorded the data of two beverages. My group chose apple juice and berry juice. The volume was 5mL for both juices and the mass ranged between 0.75g and 1.15g. The density of apple juice and berry juice was between 0.15 and 0.23 g/mL.
Once we finished, Mr. H told us to write a Conclusion/Discussion for the lab and make sure we had the graph attached to the data sheet. He also said if your group didn't finish, finish it and hand it in at the start of class tomorrow.
No comments:
Post a Comment
Note: Only a member of this blog may post a comment.